Professor Stephen Robertson is a paediatric geneticist. He treats and researches childhood disorders which have or are assumed to have a genetic basis. Understandably, he is a very busy individual. Splitting his time between caring for children in a clinical practice, teaching undergraduate and postgraduate genetics papers, and researching genetic disorders. Over the course of his research career Professor Robertson has always found himself drawn back to a particular topic, one which has incredible potential for use in a myriad of clinical scenarios: stem cells.
A stem cell is a cell which can differentiate, or change, into another kind of cell. Stem cells can be classified into two broad groups: embryonic stem cells and adult stem cells. At certain points in their development embryonic stem cells can differentiate into any cell type while adult stem cells can only differentiate into the type of tissue they're found in.
Professor Robertson's interest in stem cells is due to their potential use in healing damaged or dysfunctional brains. There are a cluster of stem cells called osteoprogenitor cells located beside the ventricles of your brain, an area called the periventricular zone. These cells are plentiful during the early development of your brain, but are all but gone by 18 months of age. This leaves us with a small window of time to mend dysfunctional or damaged brains while we have access to these cells. Currently we cannot stimulate these cells to repair the damaged infant brain, but this issue could be resolved by studying a disorder called periventricular nodular heterotopia.
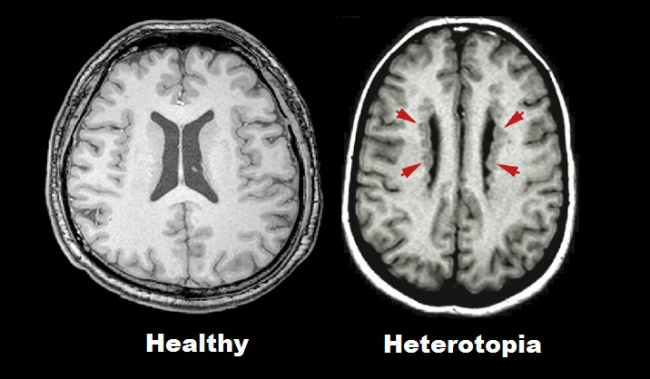
Periventricular nodular heterotopia (PH) is a disorder caused by abnormal function of neuroprogenitor stem cells. Under normal circumstances these neural stem cells would differentiate into more specific stem cells, and those stem cells would migrate away from the periventricular zone. These migrating stem cells differentiate and produce more cells as they go, resulting in all the right cells ending up in the right places at the end of brain development. In PH some of those original migrating stem cells just don't migrate. They produce functional neurons next to the ventricles, and these neurons form little lumps called nodules.
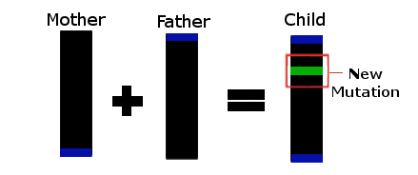
Professor Robertson and his team are examining the genes of individuals with PH to determine why these stem cells don't migrate. In order to focus on the genes which are different in PH, the researchers compare the DNA of the affected individual to the DNA of that person's parents. PH usually isn't inherited which suggests it is being caused by a new mutation in a person's DNA. By comparing the affected individual's DNA to their parents, Professor Robertson and his team can identify which genes are newly mutated and so might be at fault. In the three years they've worked on this project his team, in conjunction with a team in the US, have examined the coding DNA of more than 170 families and identified numerous genes linked to the disorder.
Why look at PH if you're actually interested in stem cells? Time and again in neuroscience we find that the best way to figure out how something works is to see what happens when it doesn't work. By looking at a disorder caused by stem cell migration failure, Professor Robertson and his team can identify some genes which are critical for its success. If that information could then be used to trigger the remaining stem cells in the periventricular zone to migrate to damaged areas, or to better control implanted stem cells, this work could have a tremendous impact on the future of neurological treatments.
While he is mindful of the potential future impact this work could have, Professor Robertson is driven primarily by the needs of affected individuals. He doesn't deny that the complexity of the issue is interesting, but asserts that curiosity should always take a backseat to human need. The needs of the patient “must always be front and centre,” he says, “that's where the need lies and where things have to be focused.”
Periventricular nodular heterotopia is not normally inherited or caused solely by environmental factors, and by the time it can be detected the nodules are already formed. For many people it has no effect, for others it causes seizures or cognitive impairments. Thanks to the work of researchers like Professor Robertson there is a chance to further categorise PH so that treatments can be more specific to those affected. While his research into stem cells is clearly important, it is his work as a clinical paediatric geneticist which has honed his focus. That focus is on the families and individuals who are looking for answers. “There is a need,” he tells me, “to step up and try to understand a family's heartache, to shine some light on their situation even if it might not be a solution.” The work he and his team are doing has tremendous opportunity to improve lives, and we are excited to see where that work takes him next.
If you've enjoyed this article, or any of the other work we do here, please consider donating to the Brain Health Research Centre. Your generosity could make a world of difference.
