Cancer biology, Department of Pathology
 Cancer is a cell growth disease where cells undergo division many more times than normal. This makes the cells prone to replication errors—mistakes that occur during the copying of the DNA on the chromosomes that occurs in each cell division. If these mistakes or mutations are not repaired they accumulate. As more and more mutations accumulate they affect cell processes involving death, location, intercellular communication and others. Eventually these rogue cells become cancers.
Cancer is a cell growth disease where cells undergo division many more times than normal. This makes the cells prone to replication errors—mistakes that occur during the copying of the DNA on the chromosomes that occurs in each cell division. If these mistakes or mutations are not repaired they accumulate. As more and more mutations accumulate they affect cell processes involving death, location, intercellular communication and others. Eventually these rogue cells become cancers.
The Department of Pathology, Dunedin School of Medicine, has a number of laboratories that work on different aspects of cancer biology.
- Cell Transformation: Braithwaite Laboratory
- Chromosome Structure & Development: Horsfield Laboratory
- Developmental Genetics and Pathology: Eccles Laboratory
- Epigenetics and Cancer: Morison Laboratory
- Molecular Oncology: Cunliffe Laboratory
- Molecular Pathogenesis: Hung and Slatter Laboratory
- Viral Pathogenesis: Hibma Laboratory
Email pathology@otago.ac.nz
Web otago.ac.nz/dsm-pathology/research/otago117838.html
Centre for Bioengineering and Nanomedicine
 Our Centre is at the forefront of research in the following areas:
Our Centre is at the forefront of research in the following areas:
- Molecular and Biomedical Imaging—advancing diagnosis and treatment of major health diseases in new ways by detecting spectral (colour) differences in tissues
- Engineered Therapeutics—triggered and controlled drug delivery systems, nanomedicine, engineered particles, selective and targeted therapies
- Regenerative Medicine—combines a patient's cells with biodegradable scaffolds and growth factors offering considerable advantages over current surgical interventions
- Bioengineering Materials—medical gels which stop bleeding, infection, and dramatically reduce adhesions and bone substitutes and bone ceramics
- Bioengineering Devices—uses engineering design principles for the diagnosis, treatment, prevention or monitoring of disease
- Medical Computing—The MPJV research group is using mobile technology to bring visibility to the patient journey; in order to deliver the right care to the right patient at the right time.
Email bioengineering@otago.ac.nz
Web otago.ac.nz/bioengineering/research
Mātai Hāora – Centre for Redox Biology and Medicine (formerly Centre for Free Radical Research)
 We carry out research on the regulation of oxidative stress and redox metabolism in cancer cells, and how these pathways may be manipulated to assist treatment.
We carry out research on the regulation of oxidative stress and redox metabolism in cancer cells, and how these pathways may be manipulated to assist treatment.
Research Projects:
- Alterations in the redox metabolism of cancer cells during metastasis and the development of drug resistance
- Targeting the antioxidant systems of cancer cells
- Vitamin C and the regulation of cancer cell epigenetics
- Role of innate immune cells in the promotion and progression of cancer
- Regulation of cell death pathways
Email mark.hampton@otago.ac.nz
Web otago.ac.nz/christchurch/research/freeradical
Centre for Translational Cancer Research - Te Aho Matatū
 Translational cancer research bridges the gap between laboratory-based science and treatment in the clinic. We now have sufficient knowledge in the fields of cancer biology, molecular biology, and immunology to make a significant impact on the treatment and management of cancer.
Translational cancer research bridges the gap between laboratory-based science and treatment in the clinic. We now have sufficient knowledge in the fields of cancer biology, molecular biology, and immunology to make a significant impact on the treatment and management of cancer.
We're bringing about rapid improvements in cancer outcomes by addressing defined clinical problems. Our research is accelerating the development and testing of new drugs and diagnostic tools that directly assist clinicians and their patients.
Our research programmes include:
- Personalised medicine
- Inherited and environmental cancer risk
- Childhood cancers
- Diagnostic tools
- Cancer and our immune system
- Drug development
Email ctcr@otago.ac.nz
Web otago.ac.nz/ctcr
Clinical Genetics Group
 Our group identified mutations in a gene called WTX as the cause of osteopathia striata congenita with cranial sclerosis (OSCS). This is an X-linked dominant disorder characterized by the accrual of dense, sclerotic bone throughout the skeleton, most especially the skull. Males who are hemizygous for a disease-causing mutation are severely affected—not only with skeletal manifestations, but also with malformations in multiple organ systems, which are often life-limiting.
Our group identified mutations in a gene called WTX as the cause of osteopathia striata congenita with cranial sclerosis (OSCS). This is an X-linked dominant disorder characterized by the accrual of dense, sclerotic bone throughout the skeleton, most especially the skull. Males who are hemizygous for a disease-causing mutation are severely affected—not only with skeletal manifestations, but also with malformations in multiple organ systems, which are often life-limiting.
Several surprising aspects have arisen out of this discovery. For instance, WTX is a negative regulator of WNT signaling and it has also been shown to be a tumour suppressor gene, implicated in the development of Wilms tumour, a kidney cancer which primarily affects children. Ongoing work in the laboratory is aimed at disentangling this developmental disease-cancer link in addition to addressing questions around how defects in WNT signaling can cause such widespread malformations in several organ systems.
Email stephen.robertson@otago.ac.nz
Web https://www.otago.ac.nz/clinical-genetics/index.html
Haematology Research Group
 Our main area of research interest is the immunobiology of cancer and leukaemia. Our long term aim is to develop better methods for the diagnosis and treatment of malignant disease.
Our main area of research interest is the immunobiology of cancer and leukaemia. Our long term aim is to develop better methods for the diagnosis and treatment of malignant disease.
The group has considerable experience in cell biology methodology including flow cytometry, magnetic bead technology, T cell proliferation assays, cytotoxicity assays, ELISAs, cell membrane biochemistry, preparation of cell suspensions from solid tissues, dendritic cell purification, immunohistochemistry and mammalian cell tissue culture.
Currrent projects include:
- Analysis of myeloid derived suppressor cell numbers and function in patients with cancer
- Investigation of mechanisms of immunosuppression by leukaemic cells
Email judy.mckenzie@otago.ac.nz
Web otago.ac.nz/christchurch/research/haematology
Healthier Lives – He Oranga Hauora National Science Challenge
 The Healthier Lives – He Oranga Hauora National Science Challenge is a national research collaboration dedicated to achieving healthier lives for all New Zealanders.
The Healthier Lives – He Oranga Hauora National Science Challenge is a national research collaboration dedicated to achieving healthier lives for all New Zealanders.
It undertakes collaborative research aimed at equitably improving the prevention and treatment of four major non-communicable diseases:
- Cancer
- Cardiovascular disease
- Diabetes
- Obesity
Our vision
He Oranga Hauora kitenga
Aotearoa hei whenua he ōrite ngā putanga hua hauora mō te tangata, kia iti iho hoki ngā pīkauranga o ngā māuiui kāore e taea te tuku ki te tangata kē.
Healthier Lives vision
New Zealand with equitable health outcomes and a substantially reduced burden of non-communicable diseases.
Many factors impact on the health of New Zealanders, including systems that affect the whole population, cultural factors that relate to particular communities, and the genetic make-up and life experiences of individuals.
Healthier Lives' research is therefore focussed within three themes:
- Healthy food and physical activity environments
- Culturally centred health interventions for Māori and Pacific peoples
- Precision medicine and personalised prevention
Email healthier.lives@otago.ac.nz
Web healthierlives.co.nz
Mackenzie Cancer Research Group
 We are interested in the pathology of human tumours and in identifying the genetic changes that give rise to cancer. Our research focuses on the molecular regulation of tumour growth, metastasis and response to therapy.
We are interested in the pathology of human tumours and in identifying the genetic changes that give rise to cancer. Our research focuses on the molecular regulation of tumour growth, metastasis and response to therapy.
Due to our close links with clinicians at Christchurch hospital and with the Cancer Society Tissue Bank, together with our laboratory expertise, we are able to carry out investigations that span from bench to bedside.
Email pathology.uoc@otago.ac.nz
Web http://www.otago.ac.nz/mackenzie-cancer/index.html
Microbiology and Immunology
 The Department of Microbiology and Immunology has a large number of research programmes across the fields of microbiology, immunology and virology.
The Department of Microbiology and Immunology has a large number of research programmes across the fields of microbiology, immunology and virology.
Cures for infectious and autoimmune diseases, as well as non-communicable diseases such as cancer and asthma, require knowledge of the immune response and how it can be manipulated.
Professor Alex McLellan is studying the immune response to cancer, specifically the roles of T cells, NK(natural killer) cells and extracellular vesicles in antigen presentation.
Professor Ros Kemp is looking at T cell and myeloid cell subsets in colorectal cancer and inflammatory bowel disease to improve diagnosis, prognosis and treatment.
Email microbiology@otago.ac.nz
Web micro.otago.ac.nz
Oral molecular and immunopathology
 Our programme, at the Sir John Walsh Research Institute, aims to investigate the cellular, and molecular basis of oral diseases, and their treatment.
Our programme, at the Sir John Walsh Research Institute, aims to investigate the cellular, and molecular basis of oral diseases, and their treatment.
Oral mucosal diseases including oral squamous cell carcinoma are being investigated using single and double layer immunofluorescence and immunohistochemistry coupled with focused micro-arrays to determine gene expression profiles. Of major interest is regulation of the local immune response in oral mucosal lichen planus and in oral squamous cell carcinoma.
Angiogenesis, and endoplasmic reticulum stress are also being investigated in these lesions so as to develop a better understanding of the role of these mechanisms in the pathogenesis of mucosal lesions.
Email sjwri.admin@otago.ac.nz
Web otago.ac.nz/sjwri/research/oral-molecular-immunopathology
Otago Genomics Facility
 We provide expertise and sequencing resources to New Zealand researchers and industry including:
We provide expertise and sequencing resources to New Zealand researchers and industry including:
- Project design
- Library preparation
- Illumina sequencing
- Oxford Nanopore sequencing
- Nanostring nCounter analysis
The Otago Genomics Facility is an Illumina Propel-certified service provider for Illumina platforms, and is also a Nanostring nCounter analysis system core facility.
Email genomics@otago.ac.nz
Web otago.ac.nz/genomics
Otago Micro and Nanoscale Imaging (OMNI)
 Four well-established and highly respected research-expertise providers have joined together under one umbrella to offer:
Four well-established and highly respected research-expertise providers have joined together under one umbrella to offer:
All the expertise, highly sophisticated instrumentation, and stellar support remains in place, as do the existing locations of services on our Dunedin campus.
The four units have collaborated on a new website as a central repository of their capabilities and achievements.
For all you need to know about what can be offered, who can help, what services, training, and specialist expertise and advice is available, please contact us.
Email omni@otago.ac.nz
Web otago.ac.nz/omni
Pathology and molecular medicine
 Our research centers on the pathology of human tumors and identifying the genetic and epigenetic drivers of tumour development, including tumor growth, metastasis, response to treatment. We are also interested in investigating the role of the immune system in cancer and how inflammation can promote epigenetic change. Our current research activities are focused on prostate, kidney, and bladder malignancies.
Our research centers on the pathology of human tumors and identifying the genetic and epigenetic drivers of tumour development, including tumor growth, metastasis, response to treatment. We are also interested in investigating the role of the immune system in cancer and how inflammation can promote epigenetic change. Our current research activities are focused on prostate, kidney, and bladder malignancies.
Email pathology.uow@otago.ac.nz
Web otago.ac.nz/wellington/departments/pathologymolecularmedicine/research
Pharmacology and Toxicology
 In the Department of Pharmacology and Toxicology state-of-the-art technologies, including the use of in vivo disease models, are used to undertake integrative, cellular and molecular investigations aimed at understanding animal and human pathophysiology (and to delineate targets for novel drugs).
In the Department of Pharmacology and Toxicology state-of-the-art technologies, including the use of in vivo disease models, are used to undertake integrative, cellular and molecular investigations aimed at understanding animal and human pathophysiology (and to delineate targets for novel drugs).
Dr Sarah Baird's group is interested in how the supportive area surrounding the cancer cells, the stroma, develops and functions. The lab also specializes in determining cell death mechanisms.
Associate Professor John Ashton's group is currently investigating mechanisms of drug resistance to small molecule inhibitors in oncogene-dominant non-small cell lung cancer, and ALK positive lung cancer in particular. They also have an interest in modelling disease for drug testing, and are currently working on developing cancer spheroids for drug screening. In addition, his group is actively investigating the mechanism of action of the Sea-sponge derivative spiroleucettadine and the pharmacokinetics of camptothecin derivatives.
Many cancers are challenging to treat as they cannot be targeted by cytotoxics, and additionally they can acquire resistance to known cellular poisons. To solve this problem the Giles Group investigates redox active agents, a drug class that selectively destroys tumours based upon their redox status. The Group's work ranges from drug design through to the in depth pharmacological analysis of the mechanism-of-action of new therapeutics.
Email pharmacology@otago.ac.nz
Web otago.ac.nz/phal/research
Protein biochemistry, structural biology and apoptosis
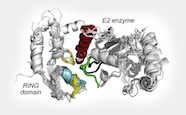 Many key events within cells are regulated by the appropriate interaction of two proteins. Understanding the molecular basis of protein-protein interactions is central to elucidation of these cellular processes and can lead to the development of new or improved therapeutic compounds.
Many key events within cells are regulated by the appropriate interaction of two proteins. Understanding the molecular basis of protein-protein interactions is central to elucidation of these cellular processes and can lead to the development of new or improved therapeutic compounds.
Professor Catherine Day is interested in understanding how a number of proteins involved in apoptosis interact, and how this knowledge can be exploited for the development of improved anti-cancer compounds. Most recently, her group has focused on characterisation of processes that result in attachment of ubiquitin to proteins because the addition of ubiquitin regulates protein interactions and protein abundance—both of which regulate signalling pathways.
Email catherine.day@otago.ac.nz
Web Department of Biochemistry – Professor Catherine Day
Research Infrastructure Centre
 Our Research Infrastructure Centre offers specialist expertise, technology and facilities in protein research, genomics, imaging, and biomedical research.
Our Research Infrastructure Centre offers specialist expertise, technology and facilities in protein research, genomics, imaging, and biomedical research.
We support optimum design and analysis of your research, and provide skill development opportunities.
Our academic leadership is of the highest calibre and we have extensively experienced and skilled staff.
Our services are available to researchers in institutions, industry, government and private companies.
Email ric.admin@otago.ac.nz
Web otago.ac.nz/healthsciences/research/ric
Structural biology of cellular signalling

Professor Peter Mace's lab aims to understand signalling networks that regulate how cells respond to stress or proliferative stimuli. Of particular interest are the mechanisms by which phosphorylation, ubiquitination and proteolysis act to control fundamental cellular processes such as proliferation and cell death.
Using a combination of structural biology, biochemistry and cell-based we aim to decipher how signalling proteins regulate these outcomes in normal cells, how signalling is disrupted in cancer, and how this understanding can be translated into more effective disease therapy.
Current projects in the lab include understanding stress-activated MAP kinase activation, and how ubiquitin regulates transcriptional networks in differentiating and cancerous states.
Email peter.mace@otago.ac.nz
Web Department of Biochemistry – Professor Peter Mace
Search the Health Sciences Expertise Database
 The Division of Health Sciences benefits from its high calibre of staff and their wide range of skills and research expertise.
The Division of Health Sciences benefits from its high calibre of staff and their wide range of skills and research expertise.
Our Staff Expertise Database provides details on University of Otago, Health Sciences staff. Each staff profile provides information on qualifications, current academic position, contact details, and a summary of research and publications.
You can search our database by keyword (eg cancer) or by name.
Health Sciences expertise database
Videos and research features
Otago Spotlight Series highlights
Professor Antony Braithwaite presented research about p53, and find out why elephants have it so good!

The gene that stops cancer (video approx 18 minutes)
The gene that stops cancer (PDF 2.8MB)
Professor Parry Guilford presented research about CDH1 and diffuse gastric cancer.
Finding cancer's vulnerabilities (video approx 10 mins)
Finding cancer's vulnerabilities (PDF 375KB)
Otago biochemistry research highlighted
Otago biochemistry research highlighted in Marsden 20th celebrations.
Troublesome Tribbles
Learn more about Peter Mace's work with a key protein involved in the development of blood and other cancers.
- TV Interview with Peter Mace (39 Dunedin Television, 4 mins)
- Researchers unlock secrets of troublesome Tribble protein (University of Otago news)


