Reducing the workload of the heart in aortic stenosis
This is a research study to find out whether commonly prescribed blood pressure medications can reduce the workload of the heart in individuals with aortic stenosis.
The heart and aortic stenosis
The human heart has four chambers which work together to pump blood around the body. Blood comes from the head and body to the heart and is pumped to the lungs. Once oxygen-rich blood returns from the lungs to the heart it is pumped to the head and body.
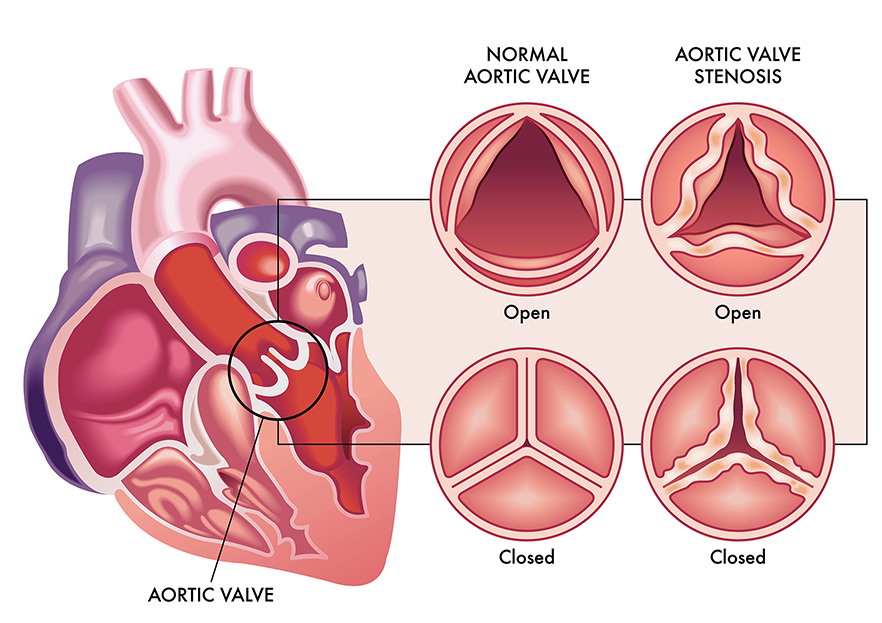
There are four flexible valves between the chambers of the heart ensuring that blood flows in the right direction. The aortic valve is the last valve that oxygen-rich blood moves through as it is pumped out of the heart, through the aorta and on to the head and body.
The flexible aortic valve can be affected by a disease called aortic stenosis. Aortic stenosis is caused by a gradual and progressive build-up of calcium in the valve, causing it to become stiff and less flexible. The increasing stiffness of the aortic valve reduces its ability to open freely when the heart beats, causing an increase of pressure inside the heart as it must work harder to pump effectively. This increased workload can potentially lead to irreversible scarring of the heart muscle.
What is the PUMAS study about?
PUMAS: PressUre reduction in Mild-to-moderate Aortic Stenosis
Currently there are no medications available to slow the progression of aortic stenosis and treatment is based on careful observation until aortic valve replacement ( AVR ), which is an invasive procedure.
AVR is typically only considered when individuals present with problems or if the heart muscle begins to fail. Even after AVR , any scar tissue remains in the heart and can increase the risk of ongoing heart related issues. We don't know if reducing the workload on the heart can alter this process, protect the heart muscle, and possibly improve the lives of people with aortic stenosis.
We are using a combination of three readily available blood pressure medicines, each with specific ways of working that may protect the heart in aortic stenosis. These medications are the calcium channel blocker amlodipine, the angiotensin receptor blocker candesartan, and the beta blocker bisoprolol.
The time it takes for aortic stenosis to progress varies, in some individuals it can take a few years for heart problems to develop, and in other individuals it can take decades. We plan to be in direct contact with study participants over an initial two-year period, and will monitor their progress after this remotely.
The investigations and procedures involved in this study are planned according to the best available medical knowledge and standards and have been reviewed by the Health and Disability Ethics Committee (reference 21/ NTA /164).
Who can join the study?
You may be able to take part if you:
- have had a heart ultrasound (echocardiogram) showing mild to moderate aortic stenosis,
- are aged between 18 and 90 years old and
- live in Otago or Southland.
You don't have to have high blood pressure to take part, and you may still be able to take part if you are normally on one of the study medicines.
What does my participation involve?
Participation in the study is entirely your choice, and includes agreeing to possibly take medicines as part of the study, and recording your blood pressure before a study visit using a home blood pressure monitor, which we will provide. We will also ask you to have blood tests taken. The study involves one visit at the hospital and your choice of further visits at the hospital, over the phone, or online.
You will have a random (50:50) chance of being allocated to continue with usual care (where we don't ask you to take any additional medicines) or taking medicines to reduce the workload on the heart.
For more detailed information about the PUMAS study, please read the Participant Information Sheet and Consent Form:
PUMAS study participant information sheet and consent form (PDF)
Our research team
 Dr Sean Coffey
Dr Sean Coffey
Consultant Cardiologist, Southern District Health Board
Senior Lecturer, Department of Medicine (Dunedin), University of Otago
 Dr Pete McLeod
Dr Pete McLeod
Consultant Cardiologist, Southern District Health Board
PhD Candidate, Department of Medicine (Dunedin), University of Otago
 Dr Kate Sneddon
Dr Kate Sneddon
Study co‑ordinator, Department of Medicine (Dunedin), University of Otago
Contact us
If you are interested in participating in the PUMAS study, or just need more information, please contact us via the form linked below, or by email or phone:
Email pumas@southerndhb.govt.nz
Tel +64 3 470 9177
Funders
 Healthcare Otago Charitable Trust
Healthcare Otago Charitable Trust
 Health Research Council NZ
Health Research Council NZ
 Lottery Health Research Grant
Lottery Health Research Grant
 University of Otago
University of Otago